APIs are chemical compounds required to develop drugs and attain the required therapeutic effect. The production of APIs is one of the most significant processes in the modern medicine because a drug would not work without an API. APIs present opportunities and difficulties. With API manufacturing, there is great potential to the business people. The opportunities to new businesses include increased global demand of cheap medicines, increased investment in healthcare infrastructure and government promoting local production.
Active Pharmaceutical Ingredients: an understanding.
It is an API that makes any drug that is pharmaceutical active. Depending on the drug, API manufacturers use chemical synthesis, biotechnology, fermentation or extraction of nature. There are those drugs that utilize APIs as single molecules and others where APIs are utilized alongside other active or inactive components.
APIs can be divided into two major categories. Synthetic APIs can be produced through controlled chemical synthesis by manufacturers, and the basis of a majority of generic drugs is synthetic APIs. They can be biotech APIs produced by biotechnological means, i.e., fermentation, cell culture or genetic engineering and their application in vaccines, biologics and monoclonal antibodies is essential to promoting patient health. All forms of API are stakeholders in the healthcare system, in addition to offering business opportunities to the API manufacturers.
Detailed project report
Market Analysis and Projection.
The API market in the world has been increasing steadily and it likely would keep expanding as the number of people living with chronic illnesses is rising as well as the number of people in need of healthcare increases. The industry reports indicate that the entire API market in the world is expected to reach above 350 billion dollars by the year 2030 and the compound annual growth rate will be about 7 percent. India – it is already one of the largest suppliers of generic medicines in the world, and the local API production is projected to increase nearly 10 percent annually.
The national strategies and measures stimulating the growth include the Production Linked Incentive scheme and health manufacturing self-reliance. The exogenous forces are the expiry of the patents, the pressure on the large drug companies, and the increased trend towards outsourcing services, which opens up more opportunities to the API manufactures in India and other regions which are low cost. This is therefore to the entrepreneurs, an extremely expanding, long term opportunity market.
Demand and Growth Factors
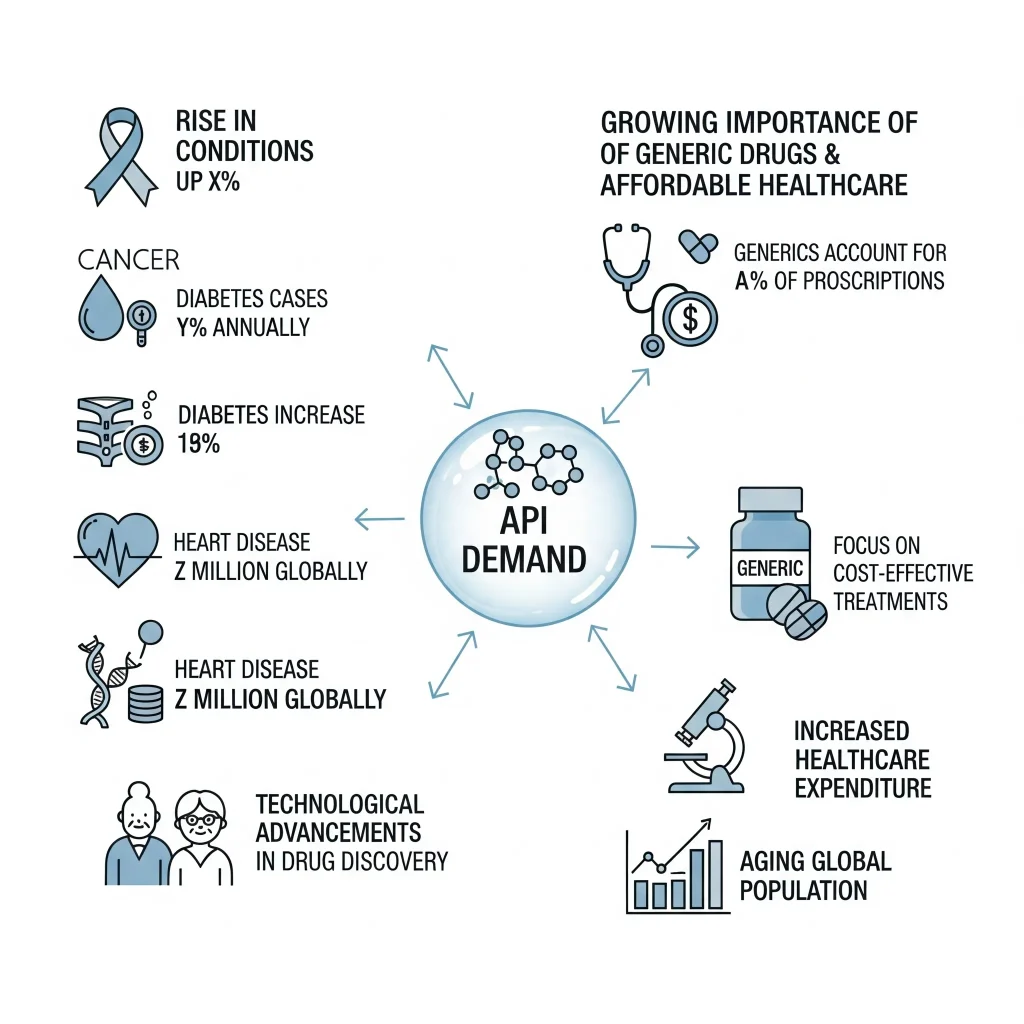
The demand in the API is a product of financial drivers and forces in the healthcare industry. One of the factors that are influencing demand is the growing rate of chronic diseases like cancer, diabetes, and heart disease, which need continuous treatment and lead to the continuous demand of medicines. Another important factor is the emergence of generic drugs as blockbuster drug patent lapses. The market of generic drugs and their API is more than ever because governments around the world are eager to explore affordable solutions to the issue of health care.
Biotechnology has, and still has been, the cause of increased demand of biological APIs needed in the current treatments like immunotherapy and highly advanced vaccines. Moreover, demand of API is also motivated by the outsourcing of API by large pharmaceutical firms, which is often outsourced to big manufacturers in India and other Asian countries in order to come up with cost effective API. All these points are indicative and reflective of the sector having very high and sustainable long-term growth.
Articles similar to this one:- Azithromycin, Cefixime, Telmisartan, Diclofenac Sodium, Aceclofenac Manufacturing. Manufacturing of Active Pharma Ingredients (API).
Regulatory Framework
Since APIs are the key components of medicines, their production is strictly controlled in most countries, not to mention all. Consumers demand that goods are produced as Good Manufacturing Practices would facilitate the production of their products in uniform, safe, and effective ways. Entrepreneurs can even be required to obtain a local regulatory approvals by Food and Drug Administration (FDA), European Medicines Agency (EMA), or Central Drugs Standard Control Organization (CDSCO) in India depending on their export markets.
They will definitely be required to meet either national or international standards of pharmacopeia (USP, BP or IP). In addition to the quality of the product, other aspects that should be included in the equation are the environmental regulations, as chemical synthesis will in most cases result in waste, which will have to be considered in relation to how to deal with. In the initial stage startups, these issues are of paramount concern to consider at the start and conduct due diligence because most times the development of credibility and access to foreign market relies on adherence to local or global regulations.
The APIs manufacturing Process.
Once synthesis of the parent molecule has been done, they recognize and purify the compound to eliminate the unwanted materials and obtain pharmacopoeial material specifications. They normally apply crystallization, chromatography, and distillation during this part of the process. After they clean the compound, they dry and mill the chemical to the right physical characteristic and a particle size to formulate.
Once synthesis of the parent molecule has been done, they recognize and purify the compound to eliminate the unwanted materials and obtain pharmacopoeial material specifications. They normally apply crystallization, chromatography, and distillation during this part of the process. After they clean the compound, they dry and mill the chemical to the right physical characteristic and a particle size to formulate.
The Quality Control team then tests every batch of material in terms of purity, stability and bioavailability. Finally, the manufacturers pack APIs under controlled conditions and store the APIs under close environmental conditioning until the drug formulators are ready to request.
To explore more, check our books..
Opportunities for Entrepreneurs
The production of active pharmaceutical ingredients (APIs) is an ever-evolving domain with innumerable opportunities. The fading patents of blockbuster drugs will continue to bolster the demand for generic APIs globally, of which there are many manufacturers. Specialty APIs, which support sophisticated therapies in oncology or rare diseases, are another large dollar opportunity.
An option of interest for entrepreneurs is contract (custom) manufacturing and producing APIs for established pharmaceutical firms across the globe, including an initiative to move more of the production to India and other cost-effective regions. Sustainability is on the rise, and eco-friendly API manufacturing is moving from a novelty to a recognized utility. Start-ups can also be tremendously successful by identifying the best niche of API production and concentrating on the quality of the product.
Challenges in API Manufacturing
While the industry is promising, it also presents several challenges. Meeting strict regulatory and quality standards requires continuous investment in compliance systems, a skilled workforce, and infrastructure. Fierce global competition often leads to pricing pressures, especially in generic APIs.
Environmental sustainability is another major issue, as regulators increasingly demand proper disposal of chemical waste. Despite these challenges, entrepreneurs can thrive by adopting innovative technologies, emphasizing compliance, and finding unique market positions that reduce reliance on high-volume, low-margin products.
Role of Technology and Innovation
Technology is transforming API manufacturing. Automation and artificial intelligence enhance predictive maintenance, quality control, and process monitoring.
Green chemistry and sustainable practices are reducing environmental impacts. Biotechnology and genetic engineering are creating new opportunities for producing complex APIs that traditional methods cannot achieve.
Support from Niir Project Consultancy Services
For entrepreneurs entering this complex industry, expert guidance can be invaluable. Niir Project Consultancy Services (NPCS) prepares Market Survey cum Detailed Techno Economic Feasibility Reports covering crucial aspects such as manufacturing processes, raw materials, plant layouts, and financials. These reports help entrepreneurs evaluate the feasibility of starting new businesses and empower them to make informed decisions. With this support, startups can navigate the sector’s challenges and better their chances for success.
Future of API Manufacturing
The future of the API industry is shaped by innovation, sustainability, and the global push for self-reliance in healthcare. Countries are increasingly trying to reduce dependence on imports and strengthen local manufacturing capabilities.
This creates an opportunity for entrepreneurs not only to engage in a profitable sector but also to enhance healthcare security. Those who invest in these areas will lead the next generation of pharmaceutical production.
Conclusion
Producing Active Pharmaceutical Ingredients is both a business opportunity and a significant responsibility. APIs are the therapeutic backbone of modern medicine, and their quality impacts the effectiveness of drugs around the globe. For entrepreneurs, this sector offers long-term growth potential driven by strong global demand, government backing, and technological progress.
Success in this field relies on compliance, innovation, and a steadfast commitment to quality. By adhering to these principles and seeking appropriate guidance, entrepreneurs can build sustainable ventures that are crucial to the future of healthcare.
Frequently Asked Questions
What is the primary role of an API in a drug?
The API (Active Pharmaceutical Ingredient) is the main component in a drug that produces the intended therapeutic effect. It is the biologically active part of the medicine, responsible for treating the illness or condition.
Are all APIs made through chemical synthesis?
Not all manufacturers make APIs through chemical synthesis. While this method remains the most common, some manufacturers produce APIs through fermentation using microorganisms by extracting them from natural sources such as plants or animals.
What are GMP regulations, nd why are they important for API production?
Good Manufacturing Practices (GMP) establish guidelines that ensure pharmaceutical companies consistently produce and control products according to quality standards. For APIs, GMP plays a crucial role in guaranteeing safety, purity, and effectiveness, since even a minor deviation can cause serious health consequences.
Can APIs be derived from natural sources?
Yes, manufacturers derive some APIs from natural sources. For example, they extract morphine from the opium poppy plant and obtain certain hormones from animal sources. This method represents one of the oldest forms of drug production.
What is the difference between a drug substance and a drug product?
A drug substance refers to the API, which is the active ingredient. A drug product is the final dosage form of the medicine that consists of the API and other inactive ingredients (excipients) that are mixed to form a tablet, capsule, or liquid administered to the patient.







